technology
Proprietary technology for immunoassay calibration
Badrilla has developed a proprietary technology for immunoassay calibration that upgrades a common immunoassay platform (Western blotting) to a robust quantitative platform. Quantitative Immunoassays are available in a limited number of applicatons (some ELISA assays), but are absent from the majority of immunoassay formats (Western blot, immunomicroscopy). Resarchers want to measure biomarkers accurately, reproducibly and in absolute chemical units wherever possible. To facilitate this, Badrilla has created a proprietary calibration standard technology that upgrades Western blots to a quantitative immunoassay capable of detecting biomarker concentrations in absolute chemical units (pmol/specimen or similar). This provides a route to the rapid development of simple quantitative assays for biomarkers of interest.
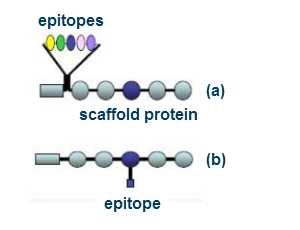
How does this technology work?
High quality calibration standards incorporate the epitope feature for a particular immunoassay into a recombinant product either by covalent attachment to a defined reactive site on a scaffold (itself a recombinant protein: b), or by manufacture as an extension to the scaffold itself a). These manufacturing approaches allow the incorporation of epitopes of a variety of complexities from simple peptides and phosphopeptides to protein domains, as required.
Calibration standards peculiar to the biomarker under investigation can be generated rapidly. Such calibration standards can be incorporated in an assay at a range of concentrations permitting the construction of a calibration curve. This can be used to determine the absolute concentration of a biomarker with high accuracy and precision.
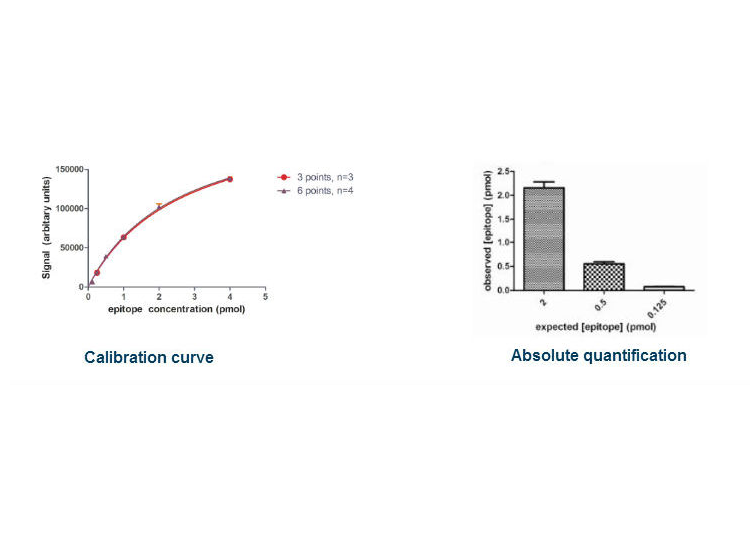
In a test system, two Western blots of a His6-epitope calibration standard were conducted. A single band of 55kDa was observed, and the signal intensity varied with calibrant loading (4pmol – 0.125pmol).
Densitometry of the raw data permits the construction of a calibration curve, which is readily defined by three calibrant concentrations examined in triplicate with exceptionally high precision. Using the calibration curve one can calculate the concentration of a biomarker examined in parallel. Observed and absolute concentration varied by less than 10%.

Which biomarkers can be calibrated?
- Linear peptide epitopes
- Post-translational modification sites (phosphorylation, glycation, others)
- Haptens (dyes, drugs, metabolites)
- Protein domains
- Single or multiple of epitopes, and
- Combinations of the above
Development through collaboration
Badrilla has conducted a program of R&D in collaboration with outstanding academic groups in the Universities of Leeds, Liverpool and Oxford. Our productive collaborative approach has allowed us to:
- Bench-mark quantitative Western blotting techniques with quantitative mass spectrometry
- Combine quantitative western blotting with high throughput western blotting to achieve higher analytical throughput
- Explore novel fabrication techniques (for the calibration standards) to extend the versatility of this technology.
Exploiting our technology
How could this technology help to deliver your research goals? Please contact us if you feel that quantitative Western blotting approaches could help meet the challenges of your business.

